500℃、20MPa时,将H2和N2置于一容积为2L的密闭容器中发生反应反应过程中H2、N2和NH3物质的量变化如下图所示,下列说法正确的是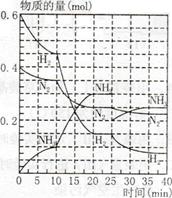
| A.反应开始到第一次平衡时,N2的平均反应速率为0.005 mol/(L·min) |
| B.从曲线可以看出,反应进行到10min至20min时可能是使用了催化剂 |
| C.从曲线可以看出,反应进行至25min时,增加了0.1mol氨气 |
| D.在25min平衡正向移动但达到新平衡后NH3的体积分数比原平衡小 |
关于CO2说法正确的是
| A.碳原子采取sp杂化 | B.CO2是正四面体型结构 |
| C.干冰是原子晶体 | D.CO2为极性分子 |
下列晶体熔化时不需要破坏化学键的是
| A.晶体硅 | B.食盐 | C.干冰 | D.金属钾 |
以下能级符号不正确的是
| A.3s | B.3p | C.3d | D.3f |
在一定温度下,可逆反应X(g)+ 3Y(g) 2Z(g)达到平衡的标志是
2Z(g)达到平衡的标志是
A.Z生成的速率与X消耗 的速率相等 的速率相等 |
| B.单位时间生成a mol X,同时生成3a mol Y |
| C.X、Y、Z的浓度不再变化 |
| D.X、Y、Z的分子数比为1∶3∶2 |
将4mol A气体和2mol B气体在2L的容器中混合并在一定条件下发生如下反应:2A(g)+B(g)  2C(g)。若经2s后测得C的浓度为0.6mol·L-1,现有下列几种说法,其中正确的是
2C(g)。若经2s后测得C的浓度为0.6mol·L-1,现有下列几种说法,其中正确的是
①用物质A表示的反应平均速率为0.3 mol·L-1·s-1
②用物质B表示的反应平均速率为0.6mol·L-1·s-1
③2s时物质A的转化率为70%
④2s时物质B的浓度为0.7 mol·L-1
| A.①③ | B.①④ | C.②③ | D.③④ |